Multiplexed Mass Spectrometry Based Cellular Thermal Shift Assay: CETSA® Explore – As A Tool For Lead Optimization
INTRODUCTION
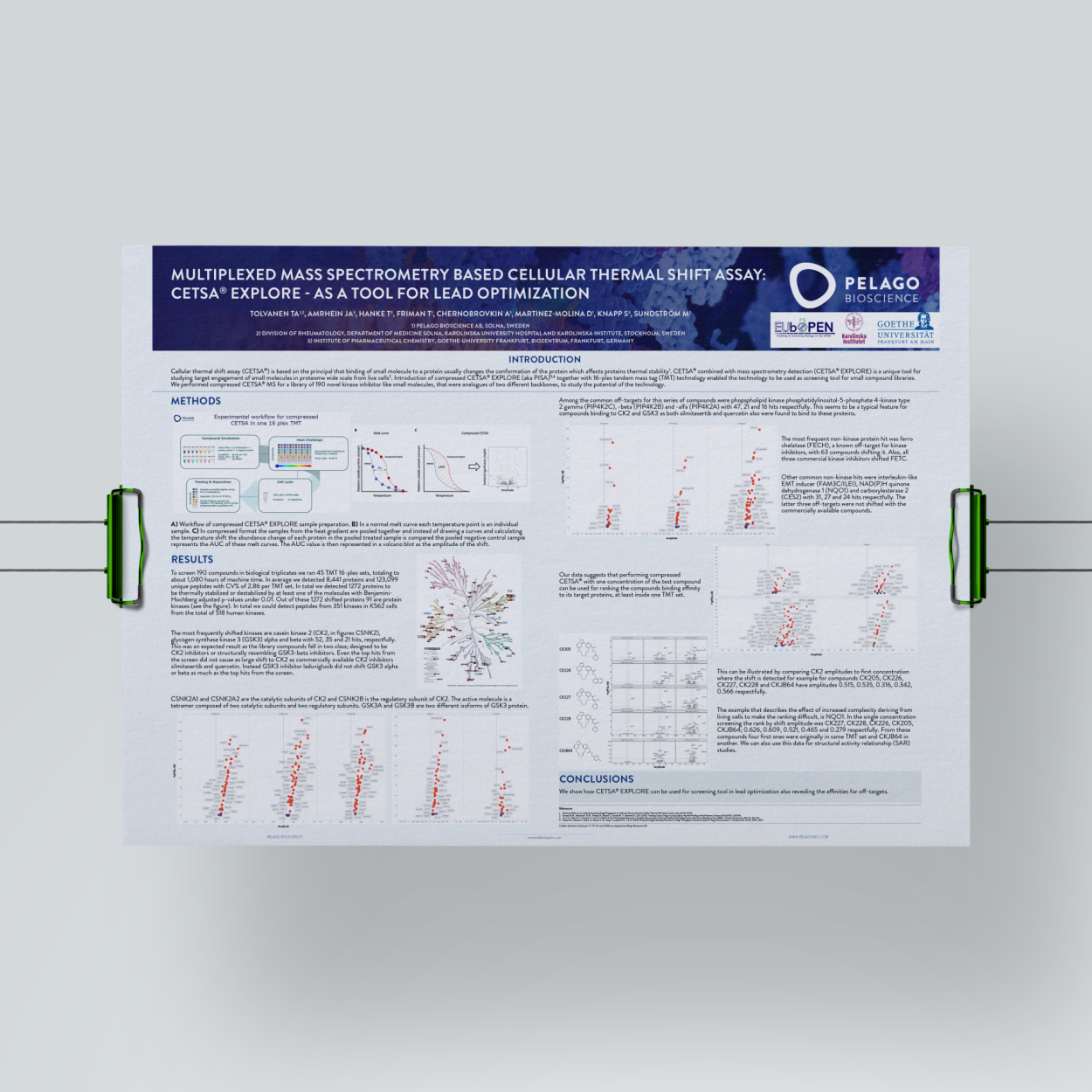
Cellular thermal shift assay (CETSA®) is based on the principle that binding of small molecule to a protein usually changes the conformation of the protein which affects proteins thermal stability. CETSA® combined with mass spectrometry detection (CETSA® Explore) is a unique tool for studying target engagement of small molecules in proteome wide scale from live cells. Introduction of compressed CETSA® Explore (as known as PISA) together with 16-plex tandem mass tag (TMT) technology enabled the technology to be used as screening tool for small compound libraries. We performed compressed CETSA® MS for a library of 190 novel kinase inhibitor-like small molecules which were analogues of two different backbones, to study the potential of the technology.

A) Workflow of compressed CETSA® Explore sample preparation. B) In a normal melt curve each temperature point is an individual sample. C) In compressed format the samples from the heat gradient are pooled together and instead of drawing curves and calculating the temperature shift the abundance change of each protein in the pooled treated sample is compared the pooled negative control sample represents the AUC of these melt curves. The AUC value is then represented in a volcano plot as the amplitude of the shift.
Key Learning
- CETSA® Explore can be used as a screening tool in lead optimization also revealing the affinities for off-targets.
How to use CETSA® Explore as a screening tool?
Publications
- Martinez Molina, D. et al. Monitoring Drug Target Engagement in Cells and Tissues Using the Cellular Thermal Shift Assay. Science 341, 84-87 (2013)
- Savitski M. M., Reinhard F. B. M., Franken H., Werner T., Savitski M. F., Eberhard D., et al. (2014). Tracking Cancer Drugs in Living Cells by Thermal Profiling of the Proteome. Science 346 (6205), 1255784.
- Liu Y.-K., Chen H.-Y., Chueh P. J., Liu P.-F. (2019). A One-Pot Analysis Approach to Simplify Measurements of Protein Stability and Folding Kinetics. Biochimica Biophysica Acta (BBA) – Proteins Proteomics 1867 (3), 184–193.
- Gaetani M., Sabatier P., Saei A. A., Beusch C. M., Yang Z., Lundström S. L., et al. (2019). Proteome Integral Solubility Alteration: A High-Throughput Proteomics Assay for Target Deconvolution. J. Proteome Res. 18 (11), 4027–4037.
